Four to six weeks. That is now the time a research team needs to design from scratch a protein capable of turning a patient's T-cells into targeted killers of their own tumor. It used to take years. The leap is not incremental: it is a different category of change.
The system that rewires the immune system
The platform was developed jointly by researchers at the Technical University of Denmark and the Scripps Research Institute in the United States. The method, published in the journal Science, demonstrates for the first time that it is possible to design proteins directly on a computer to redirect immune cells toward cancer cells through molecules known as pMHC.
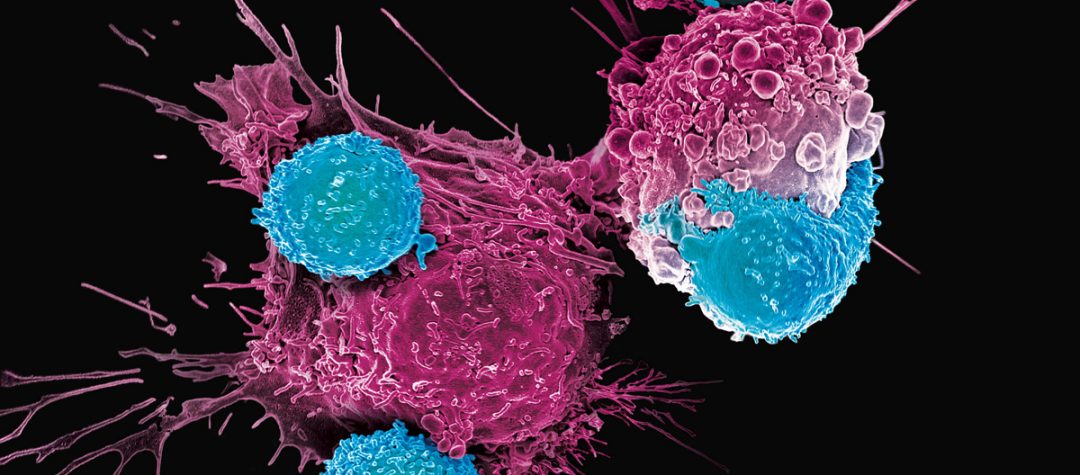
The mechanism, explained without jargon: the body's T-cells detect cancer because tumor cells present protein fragments on their surface that the immune system recognizes as danger signals. The problem is that finding the T-cell receptors that detect those specific signals for each patient is a slow, unpredictable, and difficult-to-scale process. The AI platform designs what the researchers call "molecular keys" that steer T-cells toward those tumor signals, and it does so fast enough that a new candidate molecule can be ready in four to six weeks.
IMPAC-T cells and the melanoma test
In laboratory experiments, the team tested the platform against a well-known oncological target, the NY-ESO-1 antigen, which is present in a wide variety of cancer types. The researchers designed a minibinder that bound firmly to the pMHC molecules of NY-ESO-1 and, when inserted into T-cells, created a new cellular product they named IMPAC-T cells, which effectively guided T-cells to destroy cancer cells in the laboratory.
They did not stop there. The platform also demonstrated its flexibility by designing a specific binder for a neoantigen identified in a real case of metastatic melanoma, opening the possibility of creating immunotherapies targeting tumor antigens that were previously unknown.
What sets this approach apart from the rest is not only speed. A key step in the system was the development of a virtual safety check: the platform uses AI to evaluate the designed minibinders against the pMHC molecules of healthy cells, discarding at the design stage those that might cause dangerous cross-reactions. Toxicity is filtered out before any protein reaches the lab, let alone a patient.
DTU professor and study co-author Sine Reker Hadrup summarized it precisely: by predicting and eliminating cross-reactions at the design stage, the risk associated with the designed proteins is reduced and the probability of a safe and effective therapy is increased.
The real horizon: five years to the first human trials
It is worth putting the numbers in context. The study's lead researcher, Timothy Patrick Jenkins, associate professor at DTU, estimates the method will need up to five years before it is ready for the first clinical trials in humans. The compression in design time is revolutionary. The regulatory and clinical pathway is not.
When that process concludes, the treatment will resemble in form something that already exists: patients will come to the hospital for a routine blood draw, their immune cells will be separated from the sample and modified in the lab to incorporate the AI-designed minibinders, similar to how CAR-T cells currently work in the treatment of lymphoma and leukemia.
The difference lies at the head of the process, not the tail. CAR-T requires finding existing receptors in the patient's body; this method designs them from scratch, in weeks, for any identifiable tumor target. It is not a more personalized treatment: it is a treatment built specifically for each tumor of each patient.
What was happening this week in Santa Clara
While those results were circulating through scientific communities, the Precision Medicine World Conference brought together more than 2,500 industry leaders in Santa Clara between March 4 and 6, co-organized by Stanford, UCSF, and Yale. BostonGene, whose AI foundation model for tumor and immunological biology is one of the most widely used in clinical oncology, presented data on the use of multi-omic analysis to address the complexity of cancer treatment and drug development.
Nathan Fowler, BostonGene's chief medical officer, described the central challenge of modern oncology: it is no longer the lack of data, but the ability to synthesize massive amounts of biological information into a coherent clinical strategy.
It is the same problem the DTU system is attacking, from a different angle. BostonGene acts on existing data to improve clinical decisions. The Danish team acts on the void of available therapeutic options to generate them computationally.
The conference also featured Greg Brockman, co-founder of OpenAI, as a keynote speaker on the role of generative AI in drug discovery, and Scott Gottlieb, former FDA commissioner, who addressed the regulatory obstacles to bringing innovative therapies to clinical practice. The scientific field advances; the regulatory one, at a different pace.
What the oncologist cannot do alone
There is a quiet irony in all of this. We have spent decades studying the human immune system hoping that its natural mechanisms would be enough to fight cancer. What we are discovering is that the system works, but it needs instructions that take years to find through conventional means. AI is not reinventing biology: it is reading the manuals the body already has written and generating the specific instructions it is missing for each case.
The question that no press release answers is how many patients with tumors that have identifiable targets will continue to receive standard treatments during the five years separating this Danish lab from the first clinical trials. The technology exists. The process does not, yet.
What has already changed is the design time. And with it, the very concept of what a personalized therapy means.